STAFF
Research interest
Energy insecurity and environmental pollution forces us for the development of a clean and sustainable energy technology. In this regards, fuel cell, battery, supercapacitor and solar cell have attracted much attention by the researchers. My research is focused on the design of environmentally green materials which will help in the progress for accomplishing the demand for clean and sustainable energy technology. Specifically I have worked on the design and synthesis of electrolyte for proton exchange and anion exchange membrane for application in fuel cell.
Solid Electrolyte for Proton exchange membrane fuel cell:
Graphene oxide (GO) construct a sheet-like graphene network utilizing its various polar functional groups such as epoxy, carboxylic and hydroxyl group. The ion conduction primarily occurs utilizing the epoxy groups throughout the edge and pinholes of the GO nanosheets. Although, GO shows very high proton conductivity value in the in-plane direction but the through plane proton conductivity value is comparatively low. Especially, highly stacked GO nanosheet during film fabrication using vacuum assisted method results in longer conduction track and results in low cell performance. To conquer this, we have fabricated three dimensional graphene oxide (3DGO) using freeze-drying method which represent efficient proton conduction ability and proton exchange membrane fuel cell performance. Additionally functionalization and intercalation also results in enhancing the transportation ability of the synthesized solid electrolyte.
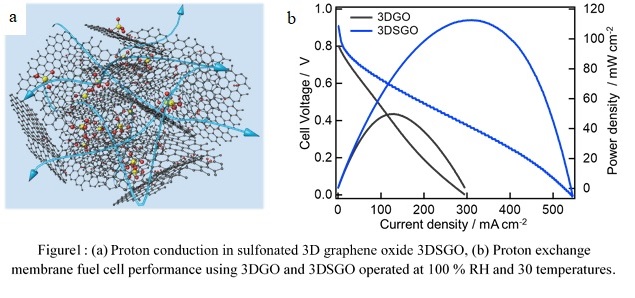
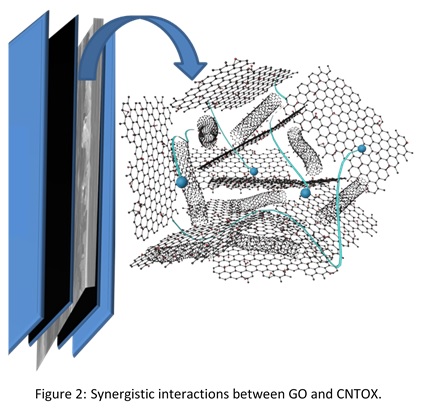
Additionally, we have established the synergy between GO and oxidized carbon nanotube (CNTOX) and both of these component acts synergistically to strengthen the structure and conducting ability in the resulting hybrid.
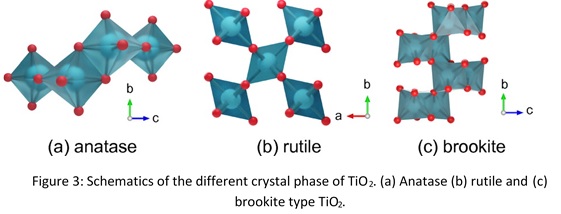
In a different study, we have also demonstrated that the specific structural arrangement of atom in different crystalline phases of a polymorph material has significant effect on their ion conduction ability. Specifically, we have synthesized titanium oxide nanosheets (TiO2) in different crystalline phases for instance, rutile, anatase and brookite having nanometer sized sheet. The difference in the proton conductivity of these crystal phases arises from the difference in their water absorption ability.
Apart from this I have worked on the design and fabrication of catalyst based on metal oxide, phosphide, sulfide, and nitride for application as catalyst for overall water splitting. Moreover, I have synthesized hybrid of metal organic framework, molybdenum disulfide, hexagonal boron nitride, layered double hydroxide, graphene oxide with metal oxide, nitride, phosphide, sulfide etc. as electrode for battery and supercapacitor. Furthermore, I have designed some carbon based electrolyte and hybrid of different layered materials for application as membrane in proton and anion exchange membrane fuel cell.
Research Activities
[Participation in conference]
- "Calcium Niobium Oxide Nanosheet for Application in Proton Exchange Membrane Fuel Cell"; M. A. Rahman, K. Hatakeyama and S. Ida, E-MRS Spring Meeting 2024, Stasbourg, France, May 27-31, 2024.
- Engineering Zeolitic Imidazolate Framework Derived Mo-Doped Cobalt Phosphide for Efficient OER Catalysts, M. A. Rahman, K. Hatakeyama and S. Ida, 18th ICC 2024, Lyon, France, July 14-19, 2024.

Achievement
Publications
- 2025
- 2024
-
- Activation of stannic oxide by the incorporation of ruthenium oxide nanoparticles for efficient hydrogen evolution reaction
- Engineering Zeolitic-Imidazolate-Framework-Derived Mo-Doped Cobalt Phosphide for Efficient OER Catalysts
- Enhanced OH− conductivity from 3D alkaline graphene oxide electrolytes for anion exchange membrane fuel cells
- Ce-doped TiO2 fabricated glassy carbon electrode for efficient hydrogen evolution reaction in acidic medium
- STAFF
-
- Wei XU
- Shinichiro Sawa
- Tetsuya KIDA
- Armando T. QUITAIN
- Junhua WANG
- Yoshihiro SEKINE
- Akira UEDA
- Yusuke INOMATA
- Muhammad Sohail AHMAD
- Jonas Karl N. AGUTAYA
- Nobleson KUNJAPPY
- Prafulla Bahadur MALLA
- Mohammad Atiqur RAHMAN
- Reetu Rani
- Kei TODA
- Tomoyasu MANI
- Agus Pulung SASMITO
- Tung Thanh TRAN
- Dario ZAPPA
- Daniel P. ZITTERBART
- Mitsuhiro AIDA
- U Rajendra ACHARYA
- Dmitri Aleks MOLODOV
- László PUSZTAI
- Yufeng ZHENG
- Gaochuang CAI
- Masahiko FURUTANI
- Hiroki MATSUO
- Zhongyue ZHANG
- Adam Karl SCHWARTZKOPFF
- Takashi ISHIDA
- Takumi HIGAKI
- Takahiro HOSONO
- Kei ISHIDA
- Makiko KOBAYASHI
- Ruda LEE
- Yuta NAKASHIMA
- Shin-Ichi OHIRA
- Atsushi SAINOKI
- Mitsuru SASAKI
- Keitaro TAKAHASHI
- Suttichai ASSABUMRUNGRAT
- Josep-Lluís BARONA-VILAR
- Nicorae BARSAN
- Jorge Norberto BELTRAMINI
- Olivier BOUTIN
- Paul BOWEN
- Pierre BREUL
- Maria Jose COCERO
- Patrice DELMAS
- Martin DIENWIEBEL
- Martino DI SERIO
- Derek ELSWORTH
- Carolina ESCOBAR
- Bruno FAVERY
- Etsuko FUJITA
- Tomonari FURUKAWA
- Jens HARTMANN
- Mohammad Abul HASNAT
- Yang JU
- Hoon KIM
- Ick Chan KWON
- Wen-Shing LEE
- Youn-Woo LEE
- Pavel LEJČEK
- Dongfang LIANG
- Bo LIU
- Tao LIU
- Hui LU
- Reiko ODA
- Yong Il PARK
- Shie-Ming PENG
- Christian RENTENBERGER
- Parasuraman SELVAM
- Amir SI LARBI
- Konstantinos Daniel TSAVDARIDIS
- Gioacchino (Cino) VIGGIANI
- Thomas WAITZ
- Yan XIAO
- Zhenghe XU
- Kazuki TAKASHIMA


